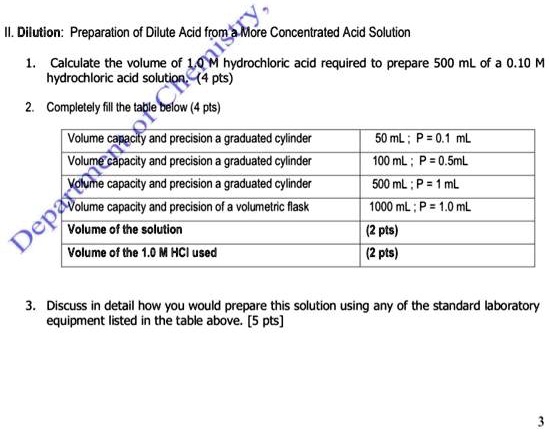
SOLVED: II. Dilution: Preparation of Dilute Acid from More Concentrated Acid Solution Calculate the volume of 1.0 M hydrochloric acid required to prepare 500 mL of a 0.10 hydrochloric acid solutlon: (4
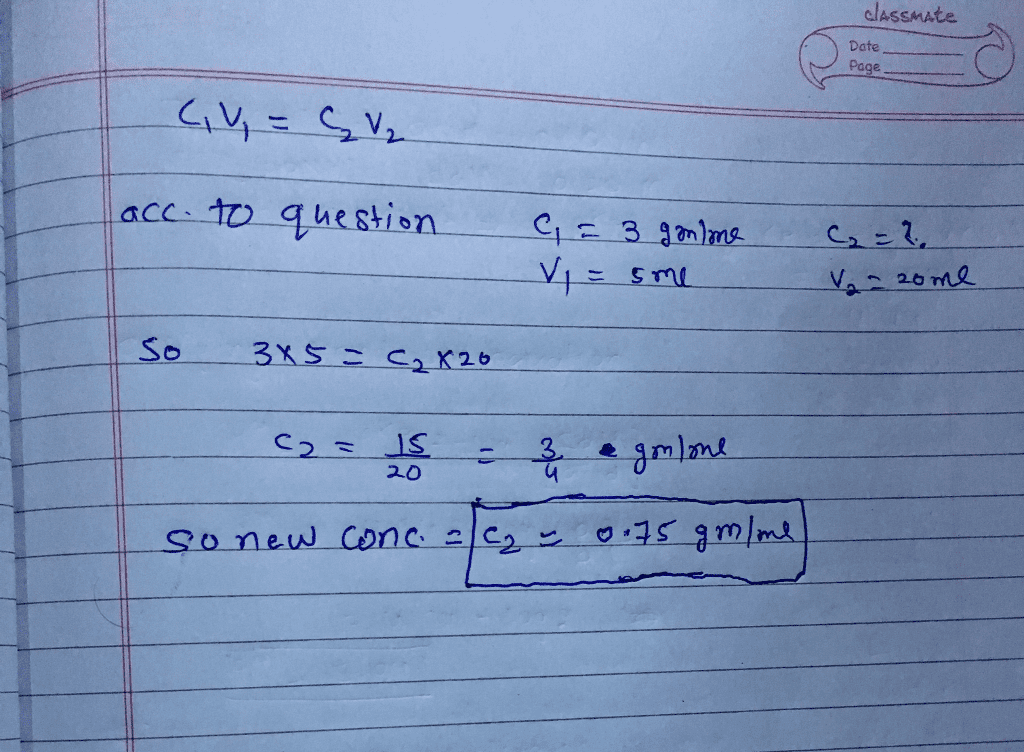
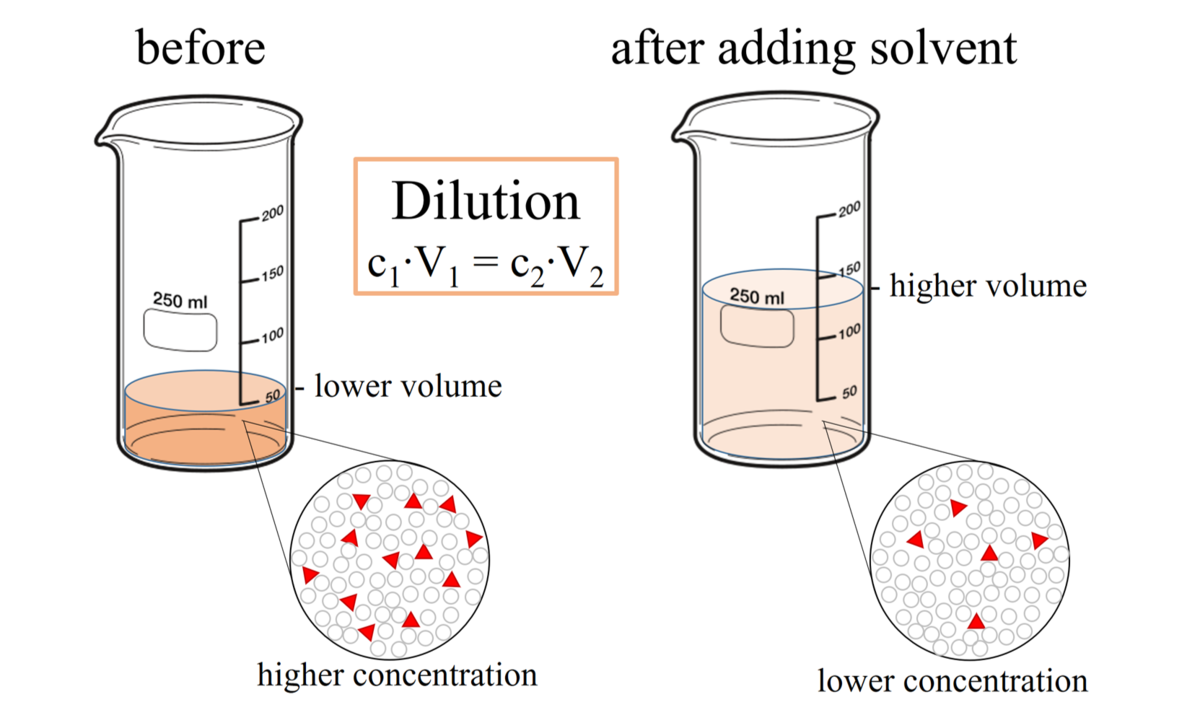
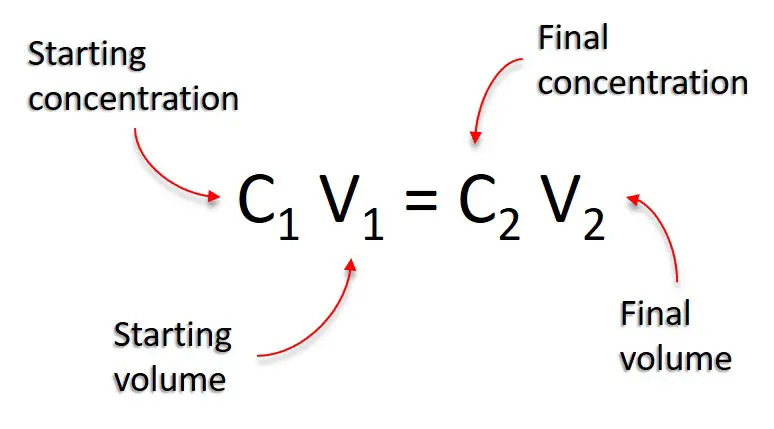
OneClass: The dilution equation is C1V1 = C2V2 where C represents the concentration of a solution and...

Brianna Bibel on Twitter: "@iubmb @ASBMB What's the bumbling biochemist's favorite equation? C1V1=C2V2! This post might have started off sounding like it would be a joke but I promise you this is