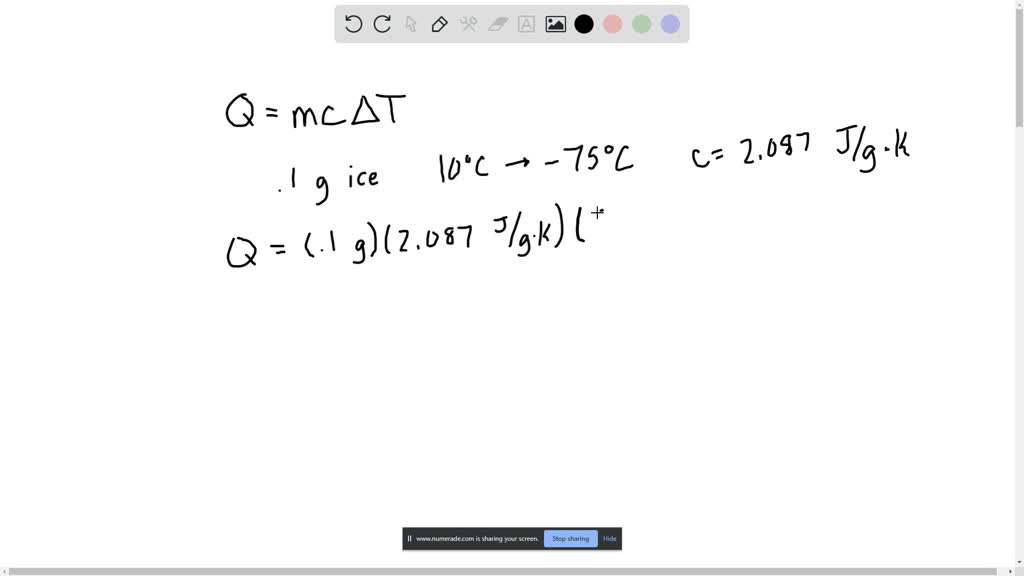
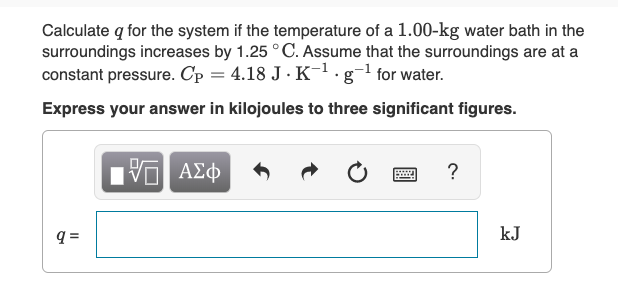
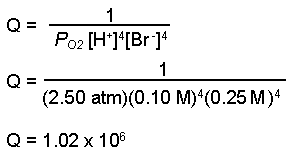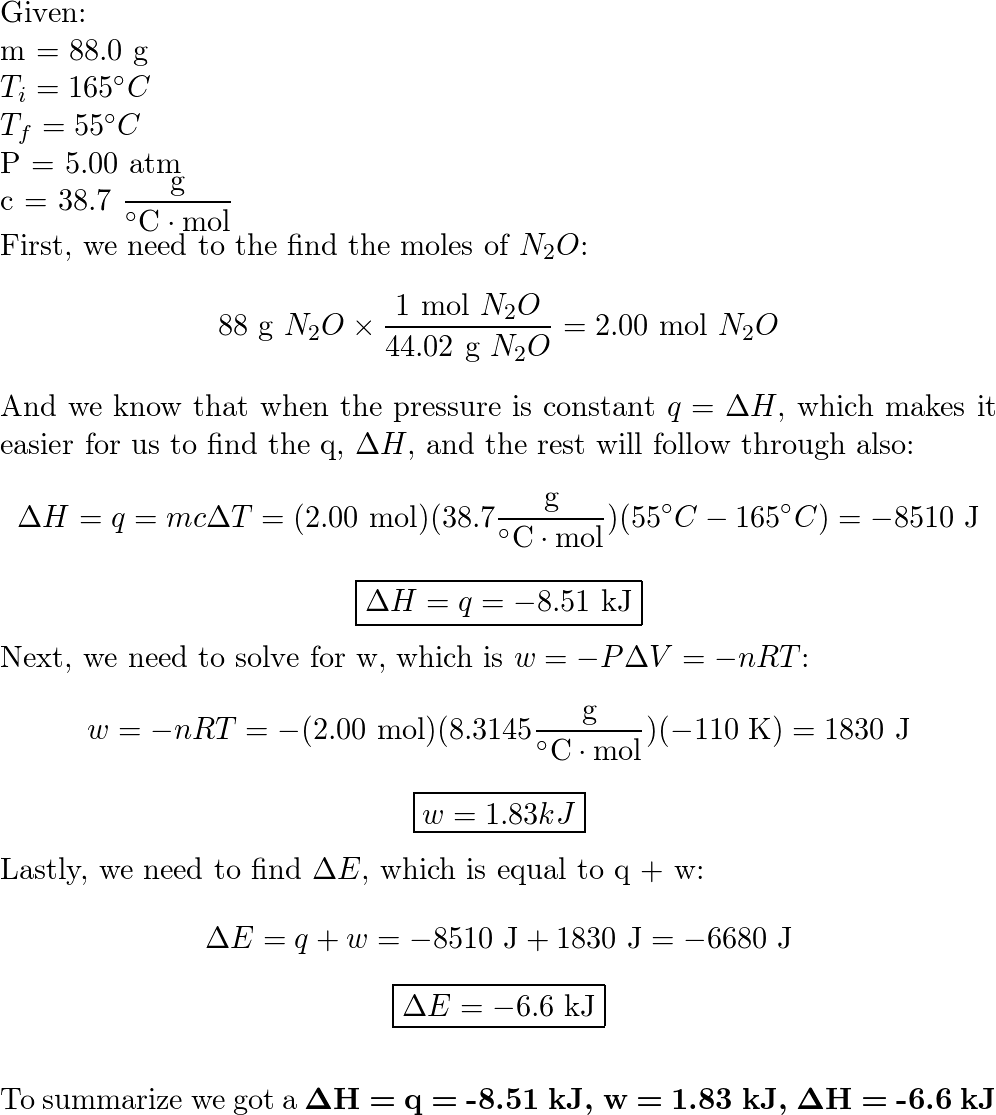Calculate q, the heat released in each reaction. Use the equation q = cmåt. (Use c = 4.18 J/g.°C and the - Brainly.com
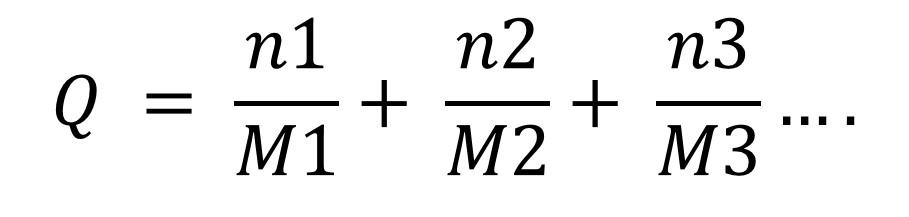
Calculating the "Q" Value for Different Dangerous Goods in a Single Outer Packaging (All Packed in One) - Daniels Training Services