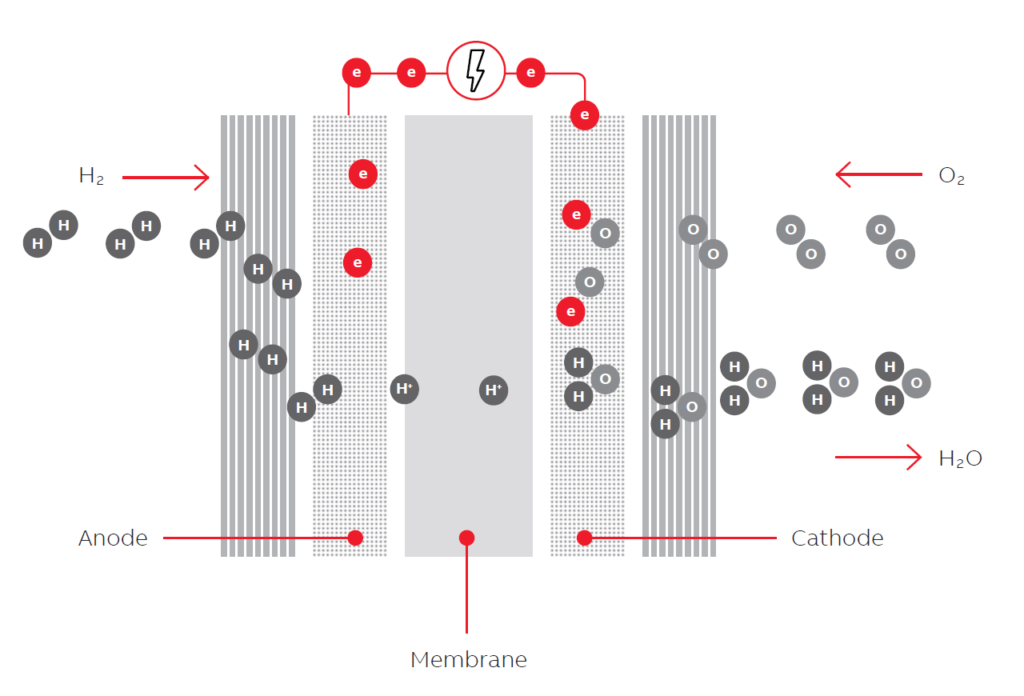
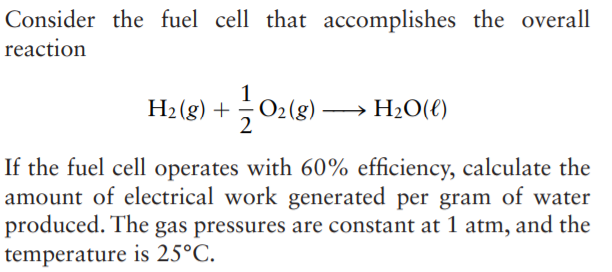How do you calculate the coulombic efficiency of a microbial fuel cell (single chamber) using high strength wastewater? | ResearchGate
Virginia Tech professor proposes simple biomass-to-wheel efficiency analysis to inform decisions on biomass/biofuel/powertrain combinations; the advantage of sugar fuel cell vehicles - Green Car Congress

Fuel cell stack and fuel cell system efficiency of the HY-LIGHT H 2 /O... | Download Scientific Diagram
![SOLVED: A hydrogen-oxygen (Hz-Oz) fuel cell operates with voltage of 0.7 V with water vapor product. [10 pts] Calculate the work (Gibbs free energy) in both kJkkmol and kWhkmol Determine the cell SOLVED: A hydrogen-oxygen (Hz-Oz) fuel cell operates with voltage of 0.7 V with water vapor product. [10 pts] Calculate the work (Gibbs free energy) in both kJkkmol and kWhkmol Determine the cell](https://cdn.numerade.com/ask_images/6caa1fcb486b465fb2c04cdf2eb0f185.jpg)
SOLVED: A hydrogen-oxygen (Hz-Oz) fuel cell operates with voltage of 0.7 V with water vapor product. [10 pts] Calculate the work (Gibbs free energy) in both kJkkmol and kWhkmol Determine the cell
![SOLVED: Q 1(c) [10 Marks] hydrogen fuel cells operates with the following reactions: Hz 2e + 2 Ht (at the anode); ^G =0 kJlmol Yz O2 + 2 e + 2 Ht SOLVED: Q 1(c) [10 Marks] hydrogen fuel cells operates with the following reactions: Hz 2e + 2 Ht (at the anode); ^G =0 kJlmol Yz O2 + 2 e + 2 Ht](https://cdn.numerade.com/ask_images/4a94a551e94645148a8ff101dcf5f8d6.jpg)




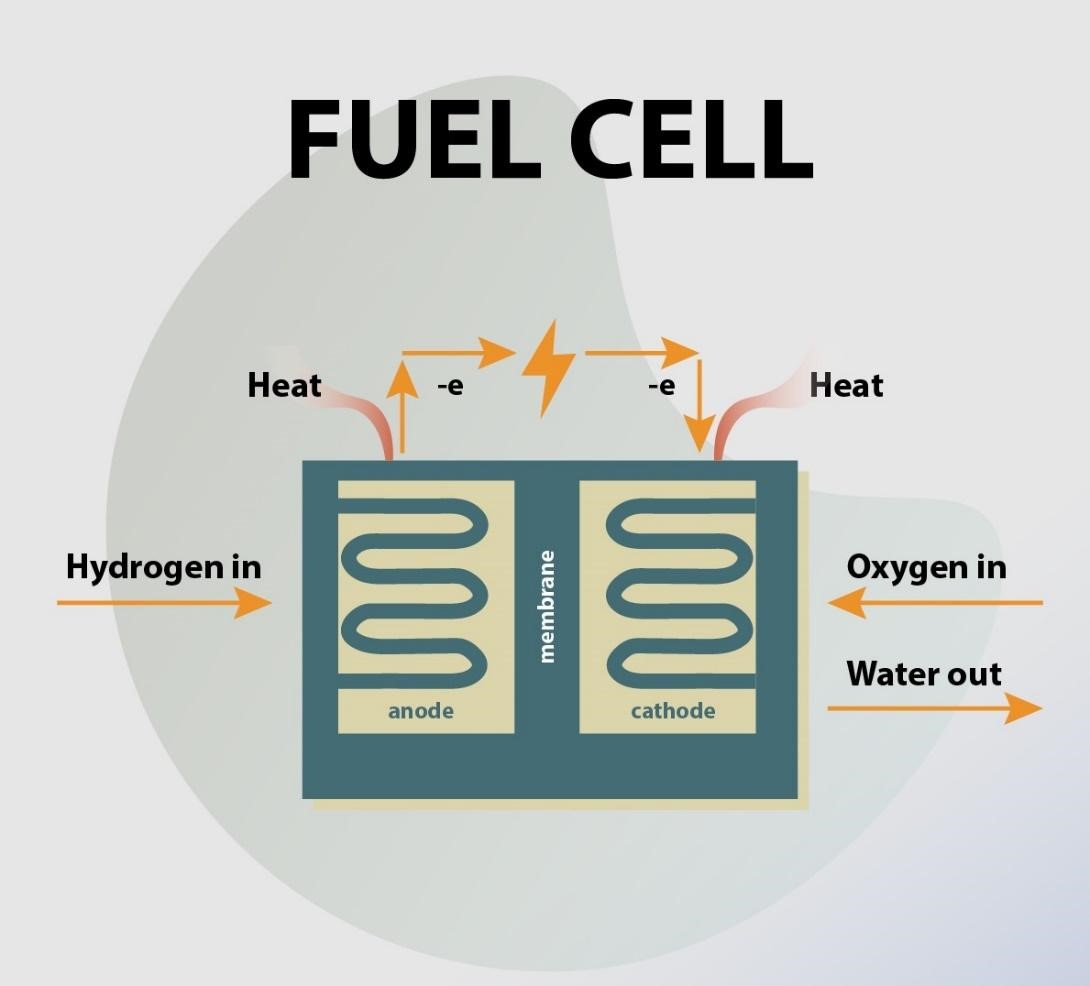

![MH-DCFC performance efficiency calculations [21]. | Download Table MH-DCFC performance efficiency calculations [21]. | Download Table](https://www.researchgate.net/profile/Andrzej-Kacprzak-2/publication/328783612/figure/tbl1/AS:690284870512646@1541588146940/MH-DCFC-performance-efficiency-calculations-21_Q320.jpg)