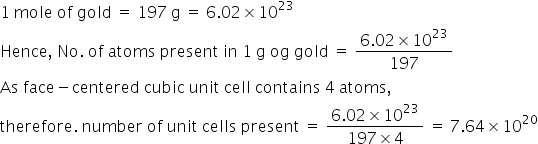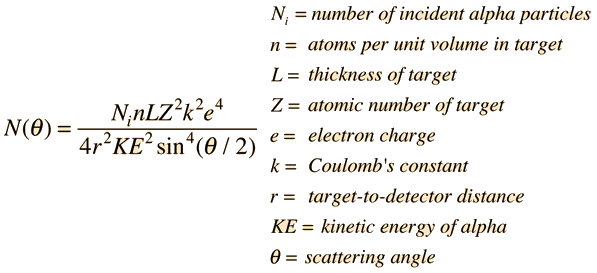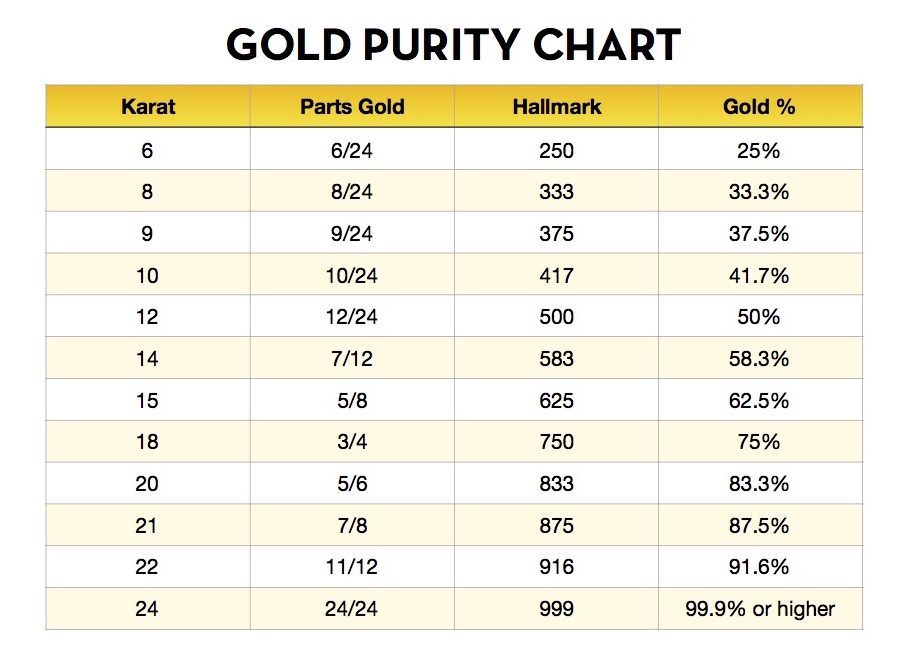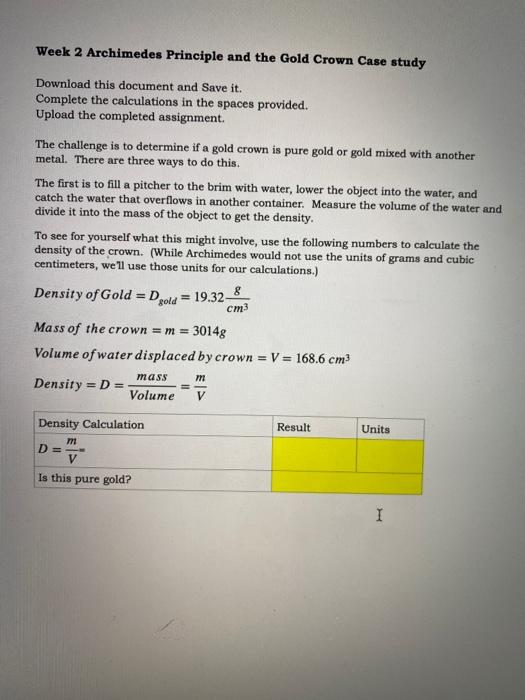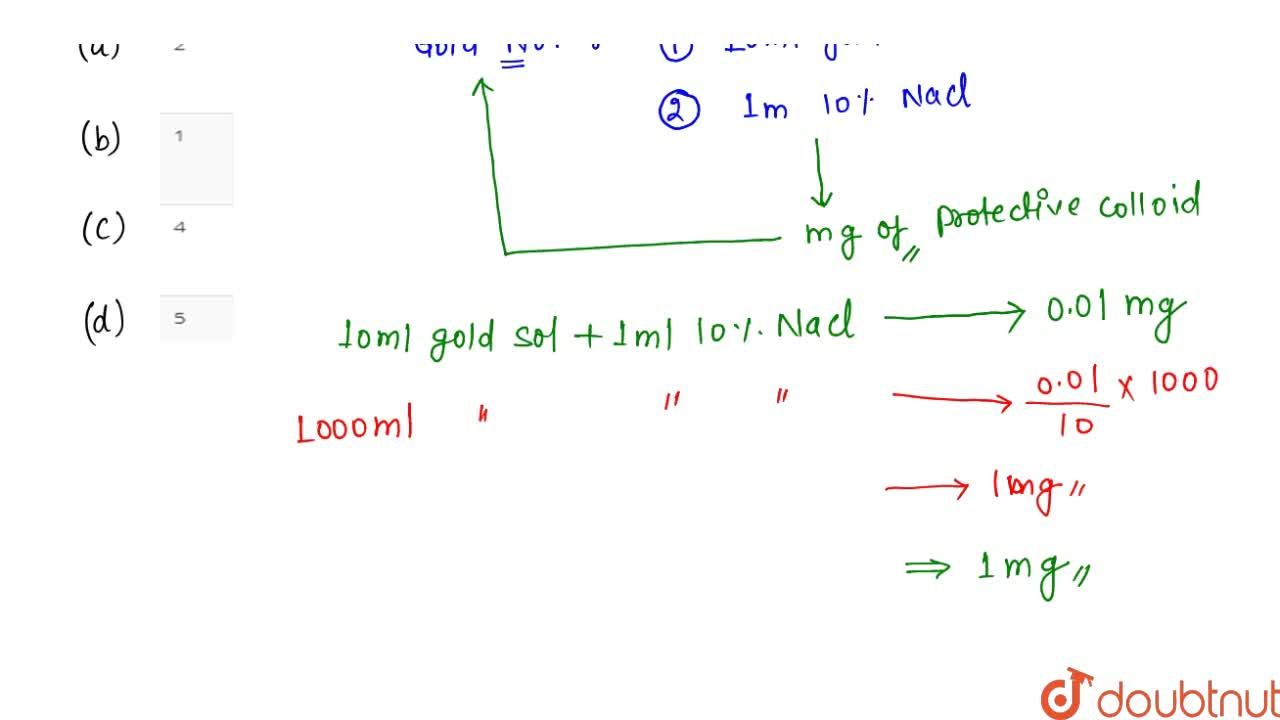
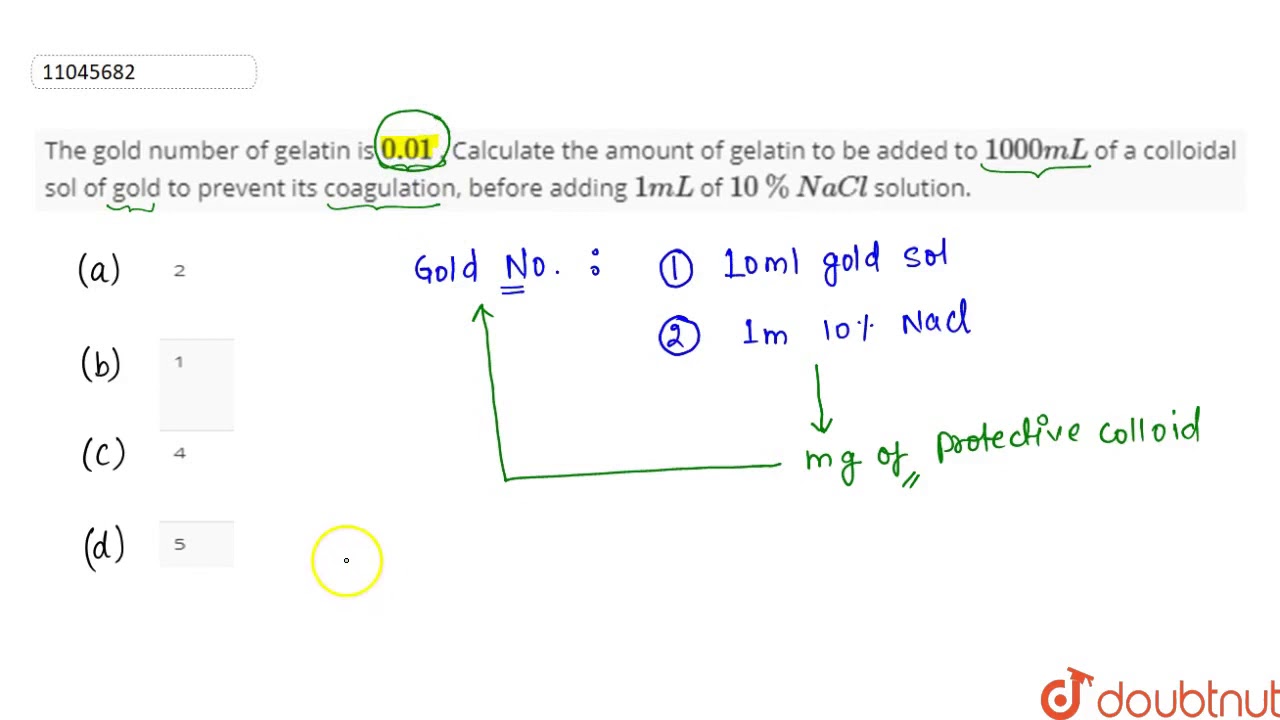
The gold number of gelatin is 0.01 . Calculate the amount of gelatin to be added to 1000mL of a colloidal sol of gold to prevent its coagulation, before adding 1mL of
The gold number of gelatin is `0.01` . Calculate the amount of gelatin to be added to `1000mL` - YouTube

Surface Chemistry Tricks to Solve Gold Number Questions Easily | Lyophilic & Lyophobic colloidal sol - YouTube

SOLVED: a) A 250-9 piece of gold is at 19 'C.5.192 kJ of energy is added to it by heat: The specific heat of gold is 129 Jkg:'C). Calculate its final temperature

SOLVED: 2.1. Svedberg gives the following table of Westgren's data for theusedi equilibrium of a gold sol under gravity: Height (um) Number %l particles 889 692 572 426 357 253 217 185