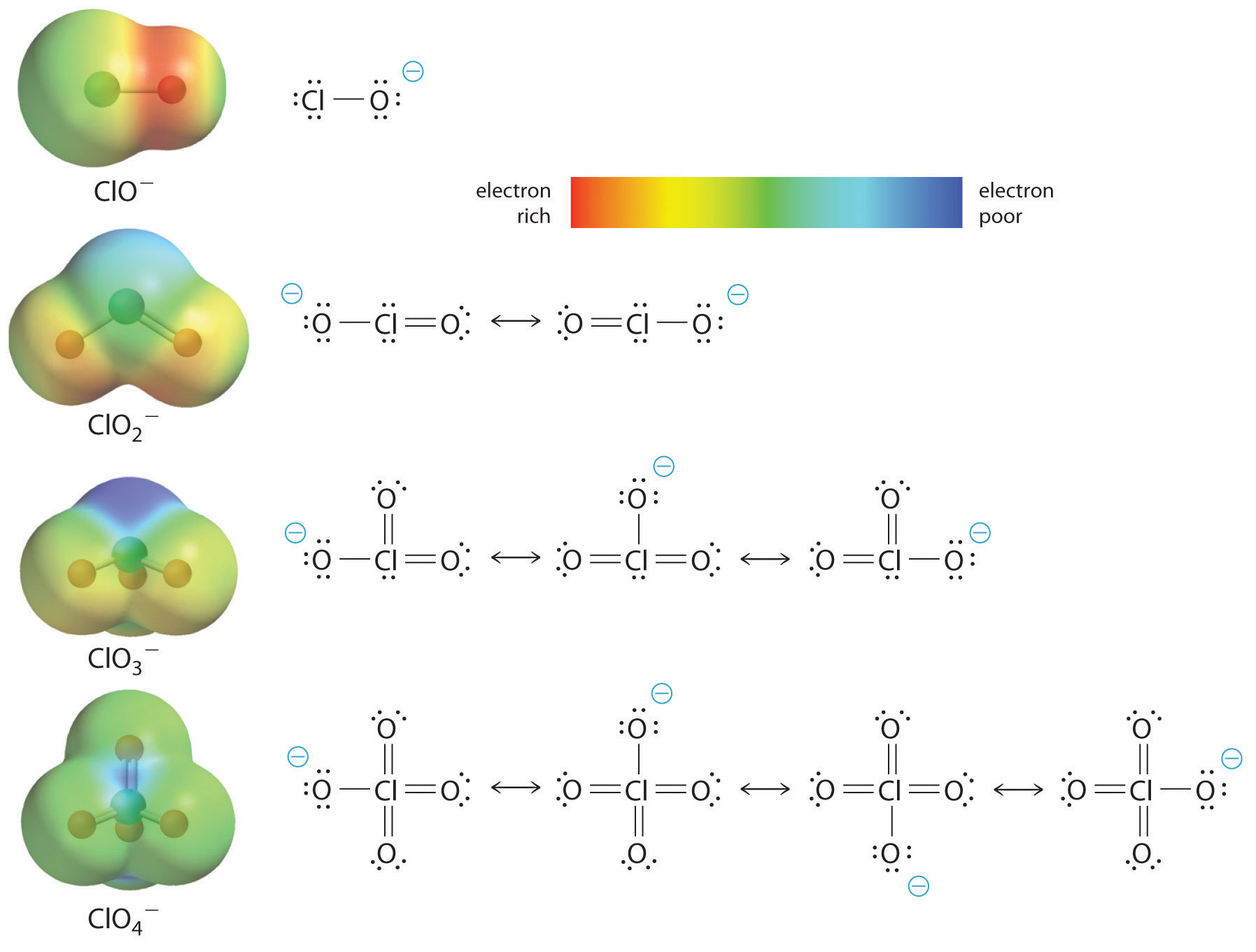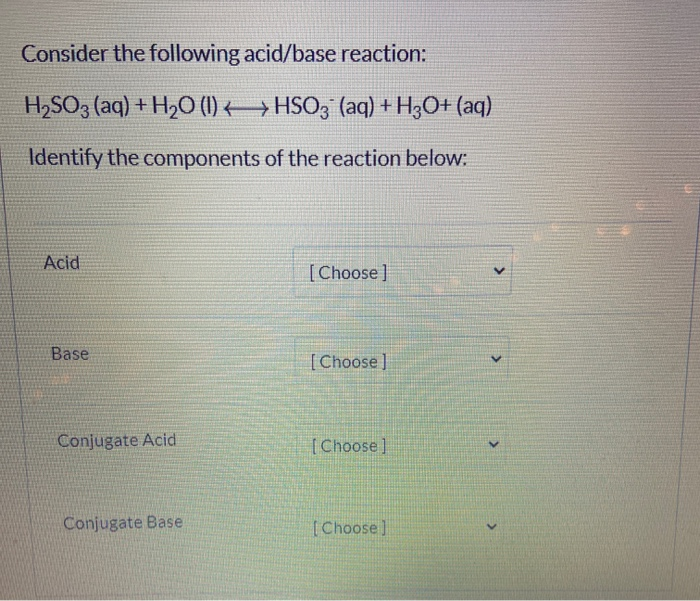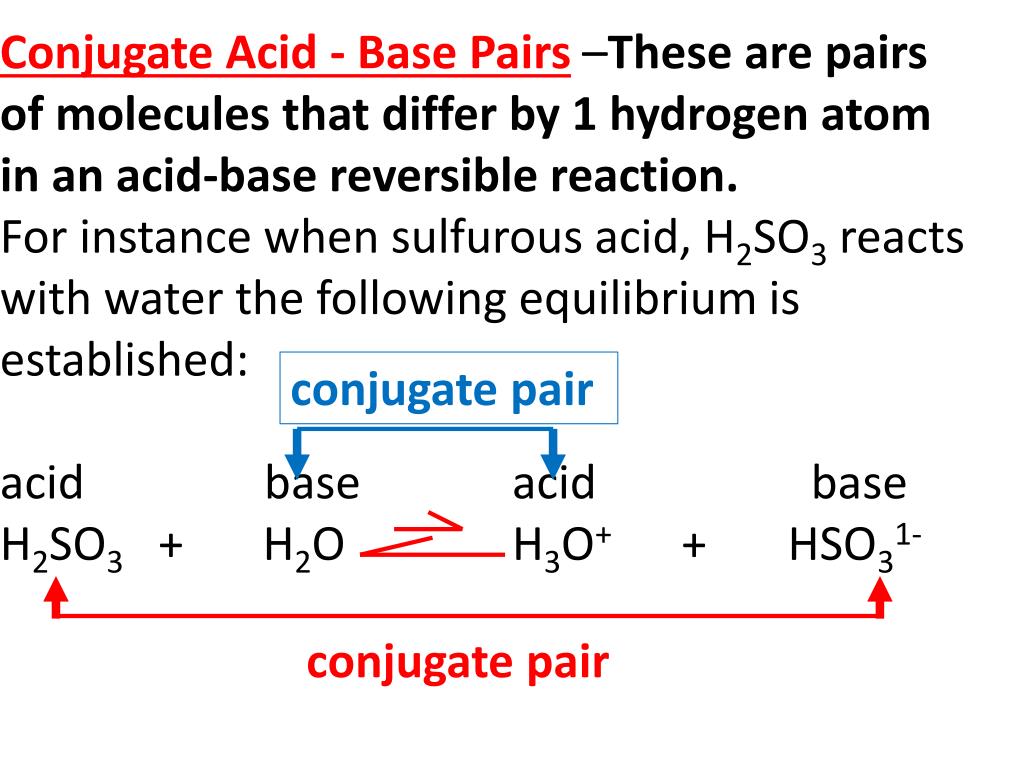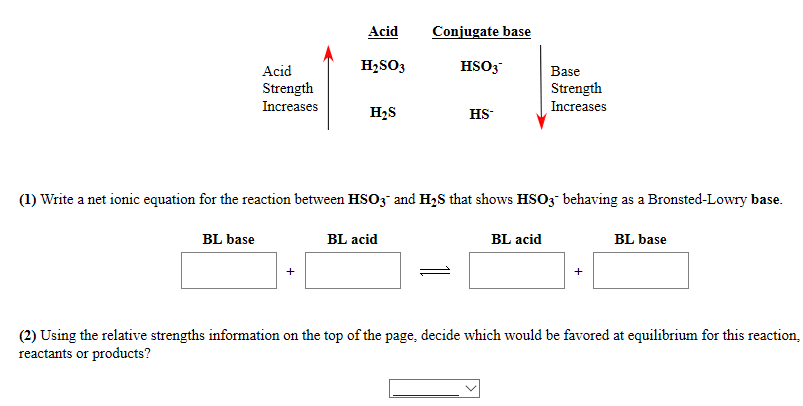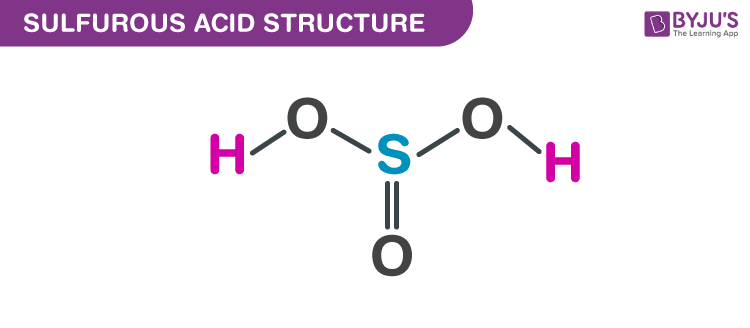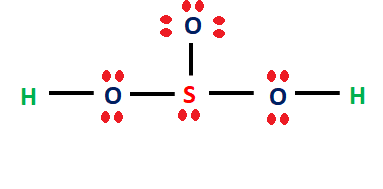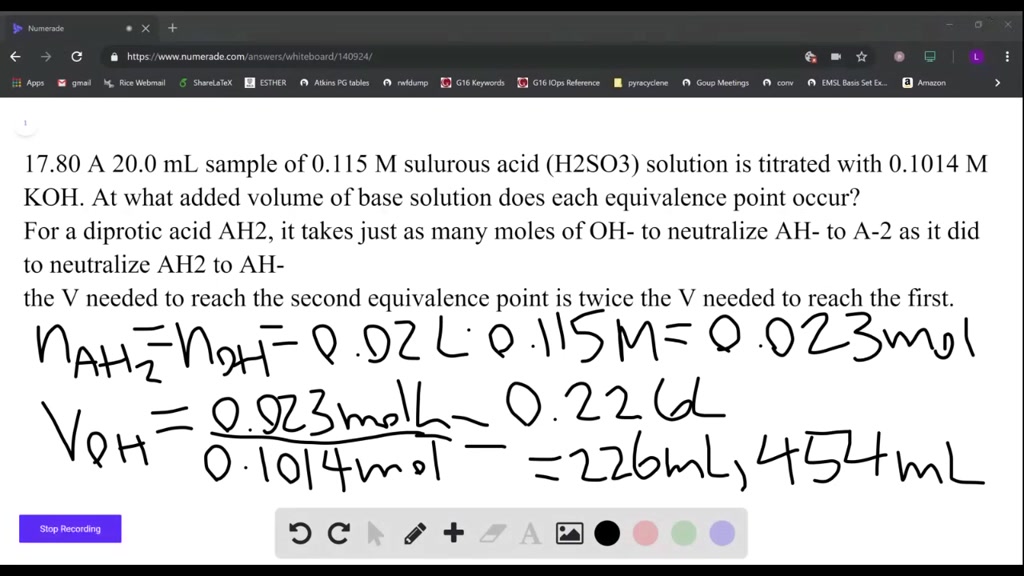
SOLVED:A 20.0 -mL sample of 0.115 M sulfurous acid (H2 SO3) solution is titrated with 0.1014 M KOH . At what added volume of base solution does each equivalence point occur?

OneClass: 1) Which of the following is not a conju gate acid-base pair? A) H2SO3/HSO3 E) None of the ...