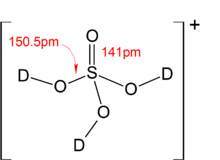
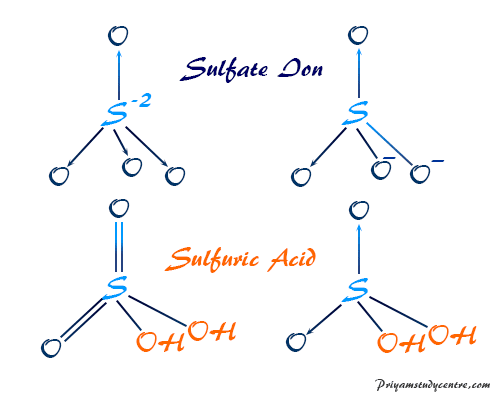
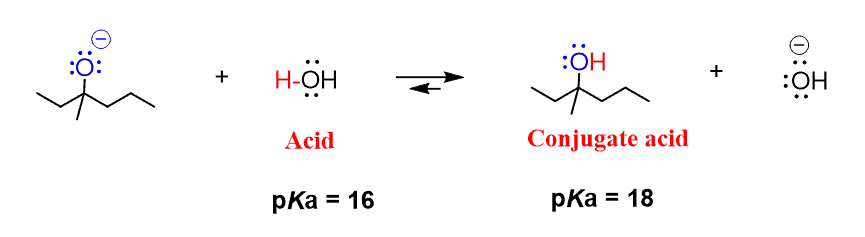
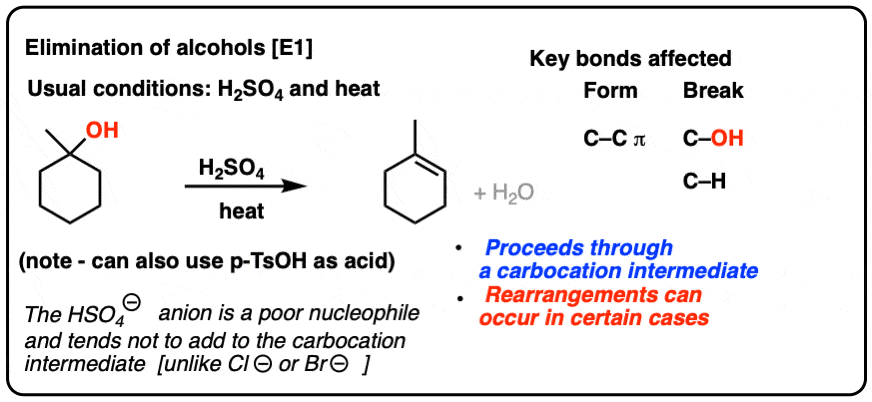
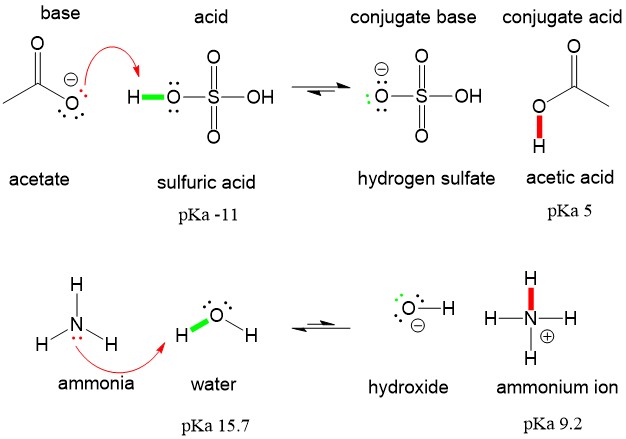
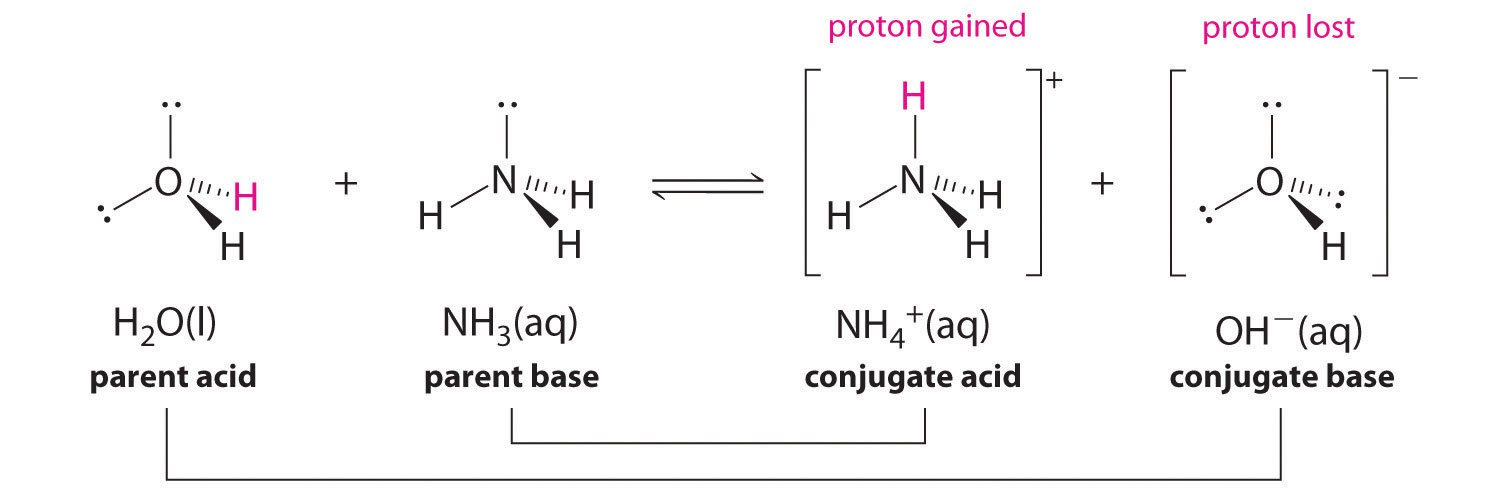
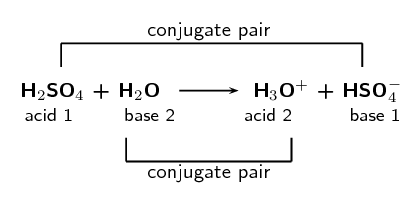
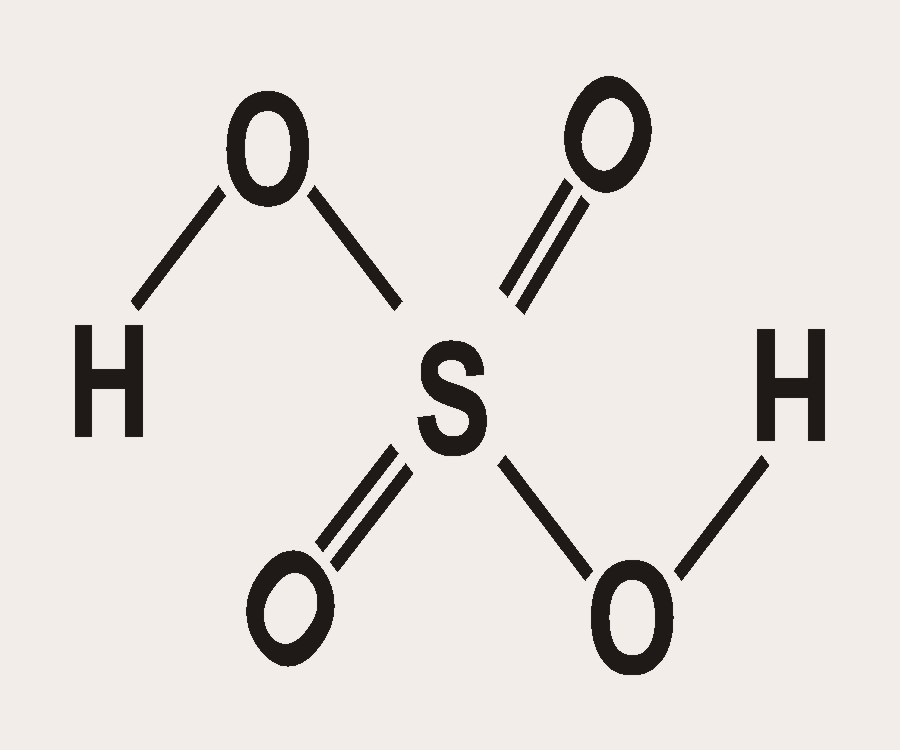
Ammonia Catalyzed Formation of Sulfuric Acid in Troposphere: The Curious Case of a Base Promoting Acid Rain | The Journal of Physical Chemistry A
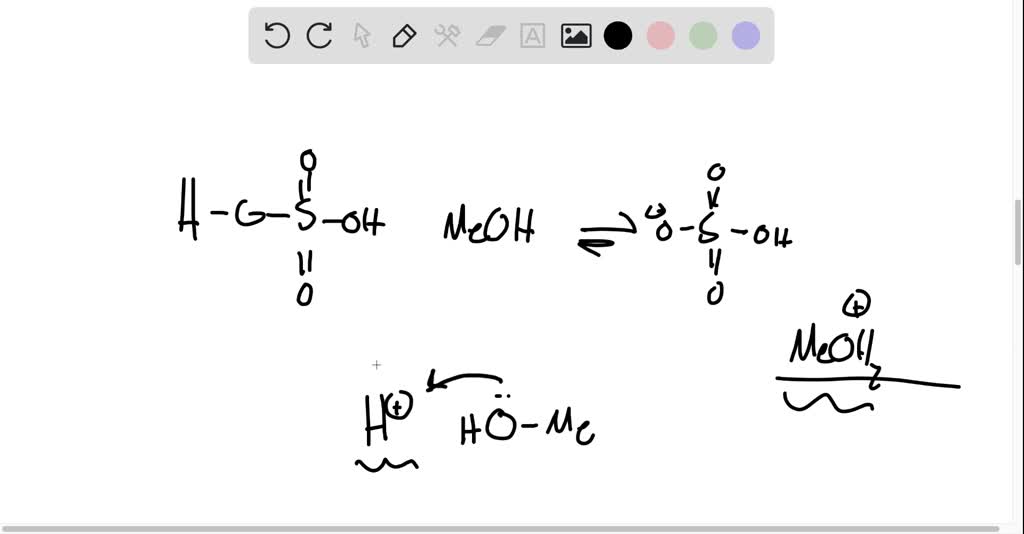
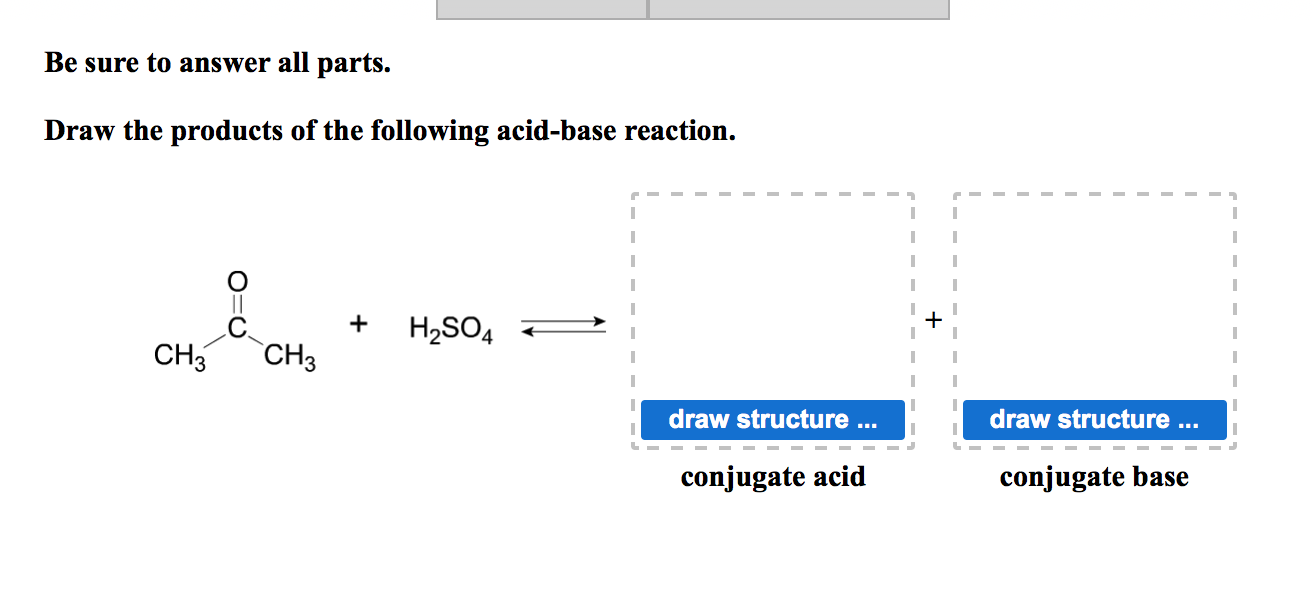
SOLVED: Organic Chemistry Write the products of the following acid-base reactions: (a) CH3OH + H2SO4 <–> ? (b) CH3OH + NaNH2 <–> ? (c) CH3NH3 + Cl- + NaOH <–> ? -