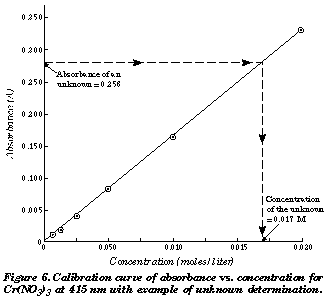
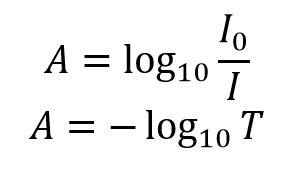From Voltage to Absorbance and Chemical Kinetics Using a Homemade Colorimeter | Journal of Chemical Education
According to Beer's Law, if the absorbance of a sample is 0.33 at a concentration of 0.20M, what is the absorbance at a concentration of 0.40M? | Socratic

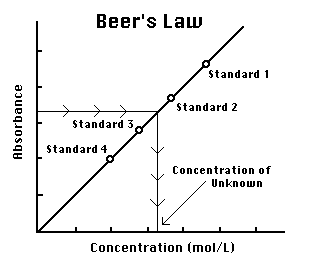
Beer Lambert's Law, Absorbance & Transmittance - Spectrophotometry, Basic Introduction - Chemistry - YouTube


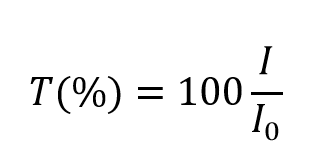


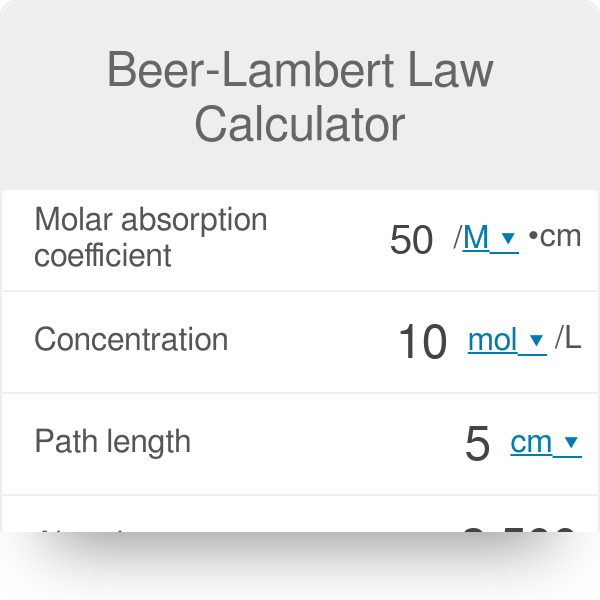

:max_bytes(150000):strip_icc()/beers-law-definition-and-equation-608172_FINAL-20ddc4fef437472db0a0ebe395770c76.png)