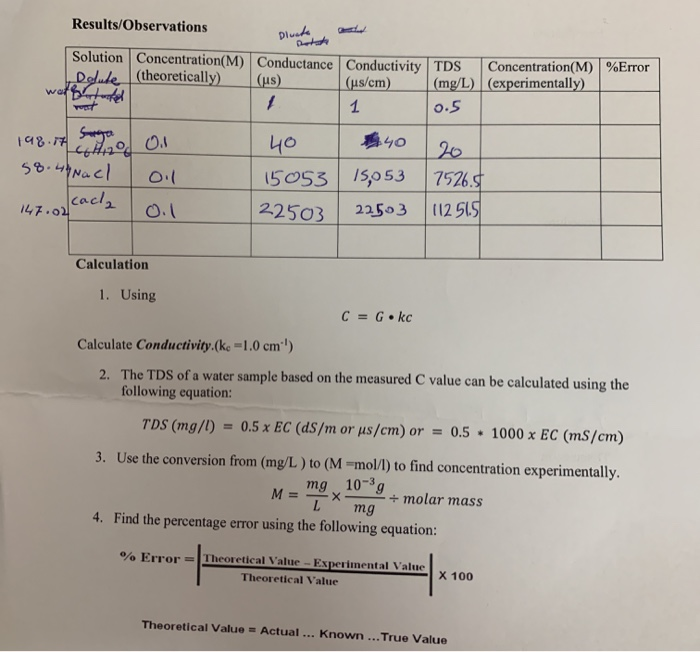
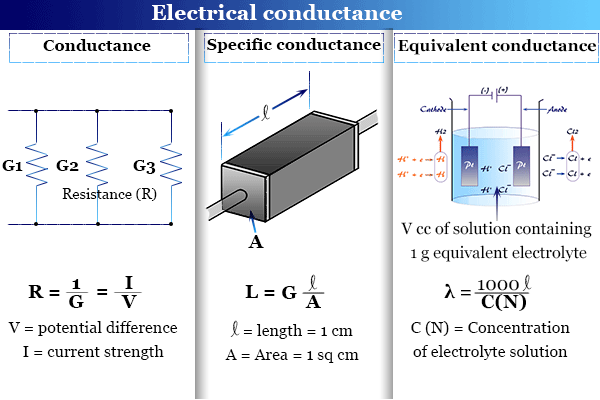
Define the following and write the formula and unit of each:(i) Specific conductivity, (ii) Molar conductivity.

Exp 4A: Conductivity Of Aqueous Solutions Purpose –Study conductivity of a series of solutions to determine the difference between strong electrolytes, - ppt download

The conductivity of `0.1`m KCl solution is `1.29sm^(-1)`. If the resistance of the cell filled with - YouTube

SOLVED: 1 We filled a conductivity cell with KCL solution of 0.0400 mol dm and its conductance at 25 %C was 2.102 mS Calculate the cell constant: The conductivity of KCL solution

The molar conductivity of a 1.5 M solution of an electrolyte is found to be `138.9 S cm^(2) mol^(-1) - YouTube

The specific conductivity of a solution containing `1.0g` of anhydrous `BaCI_(2)` in `200 cm^(3)` of - YouTube