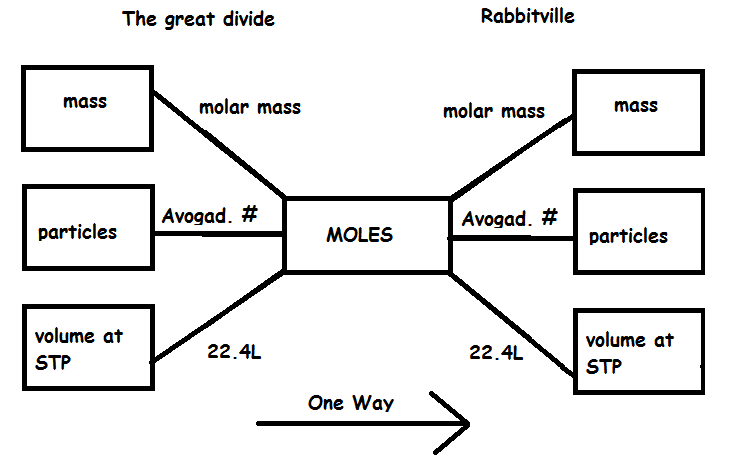
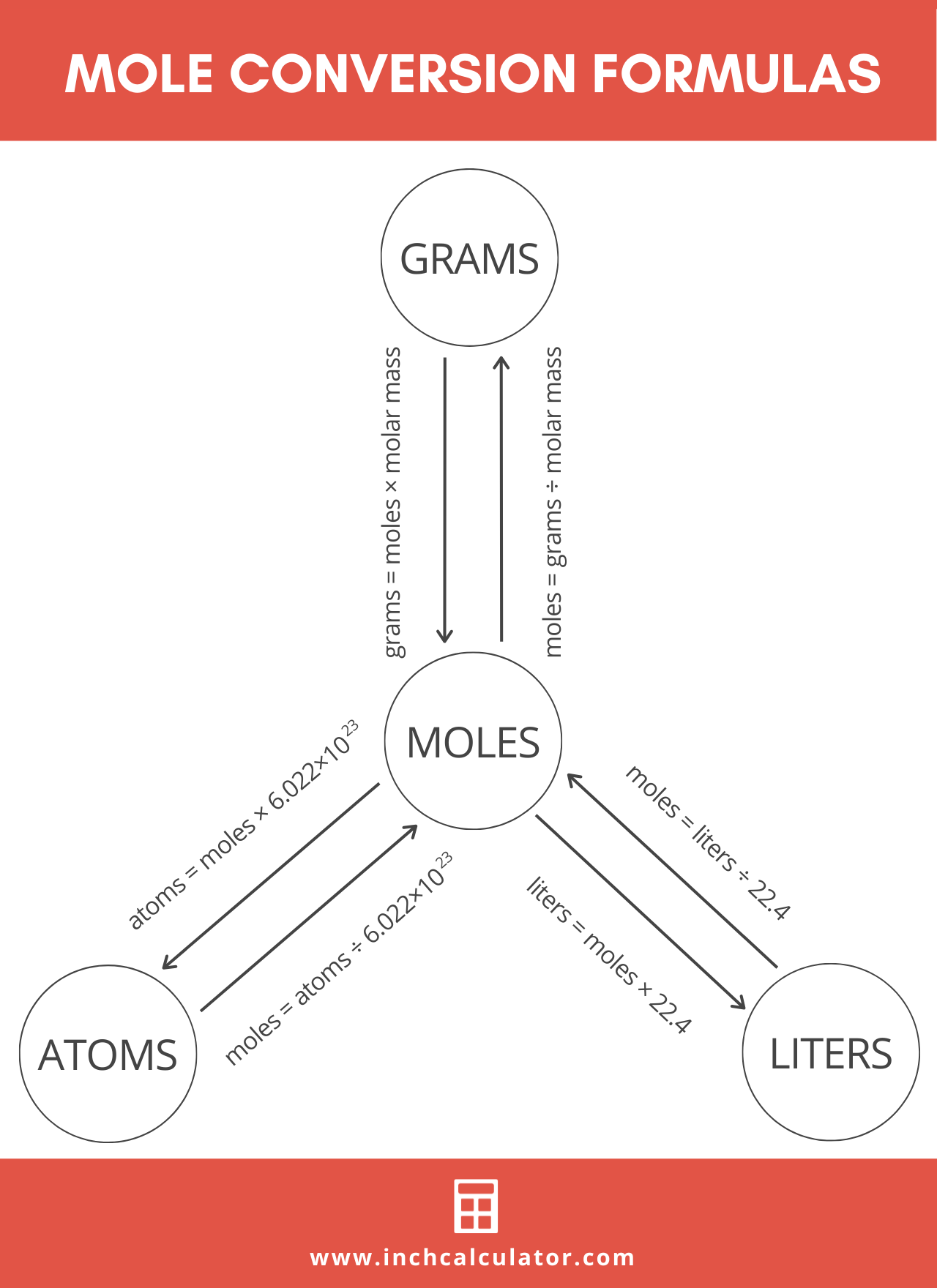
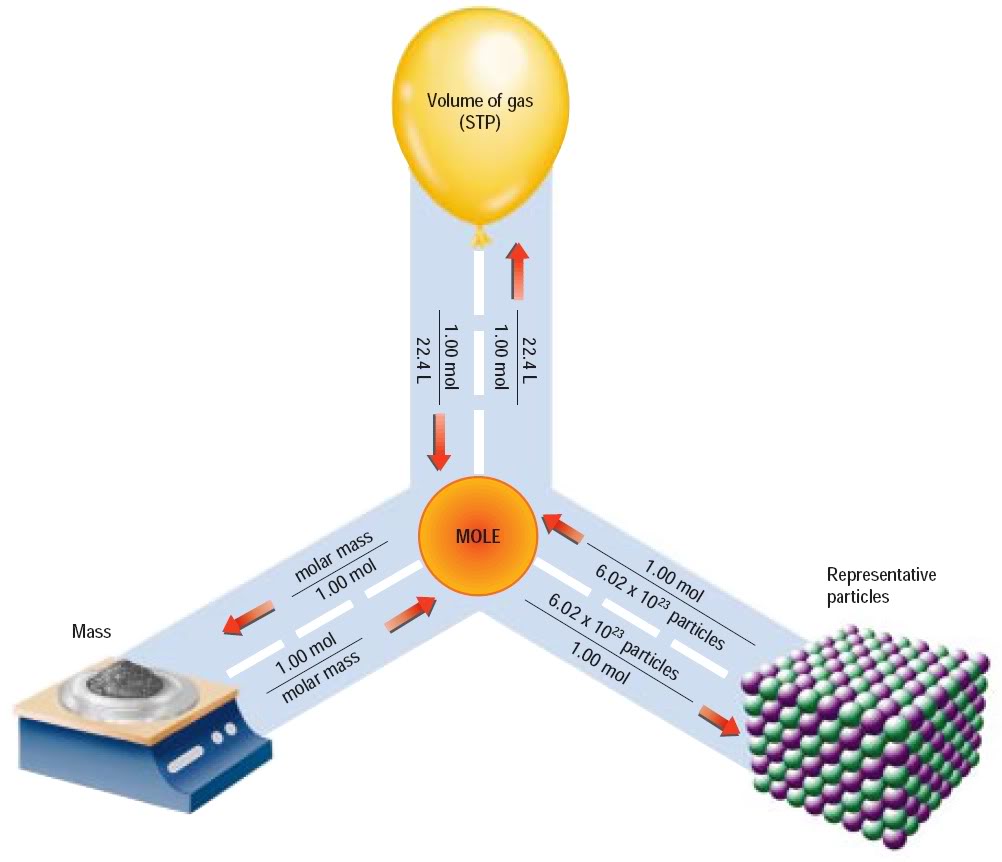
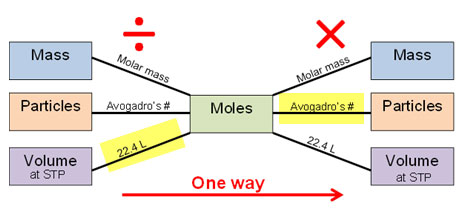
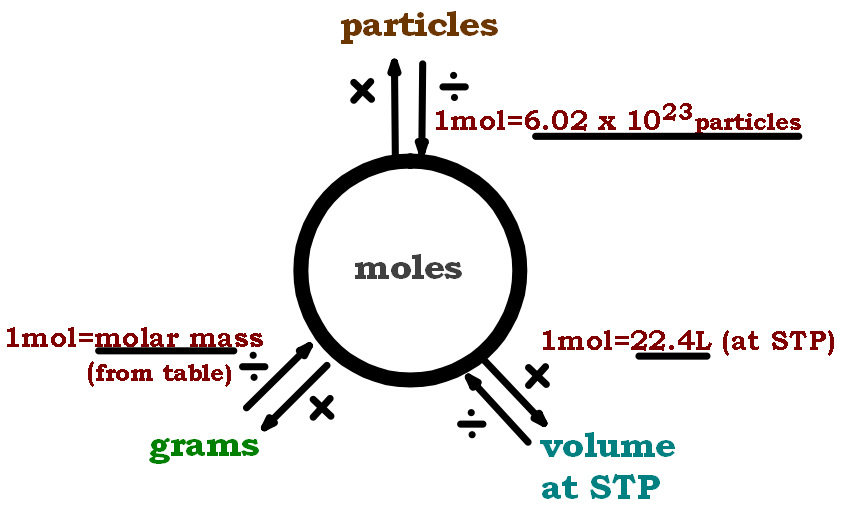
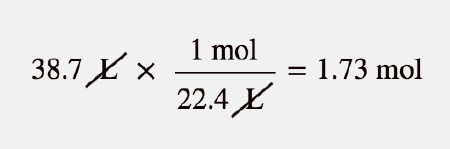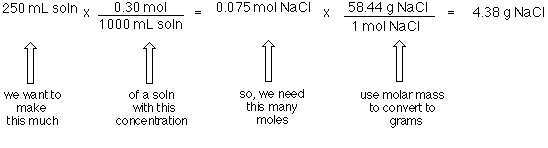SOLVED: Mole-Volume Conversions Determine the volume in liters occupied by 0.030 moles of a gas at STP. How many moles of argon atoms are present in 11.2 L of argon gas at

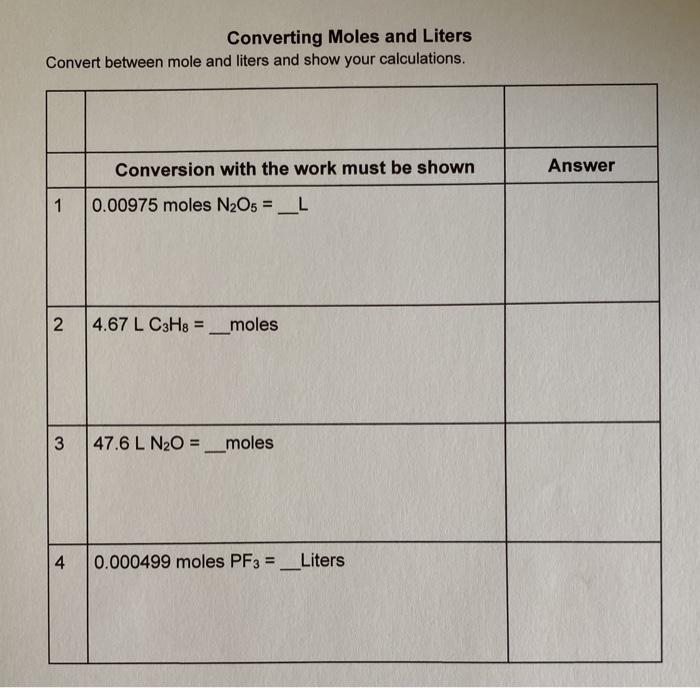
Answer Key Moles & Liters volume .docx - Moles & Liters Volume We've established that it's not practical to count atoms so we often need to use | Course Hero