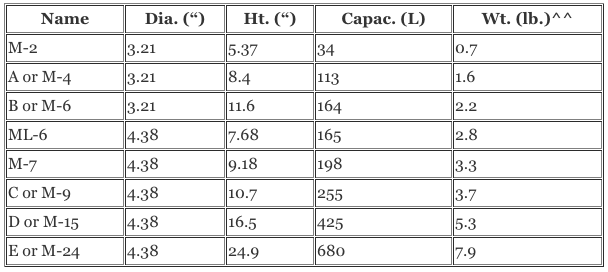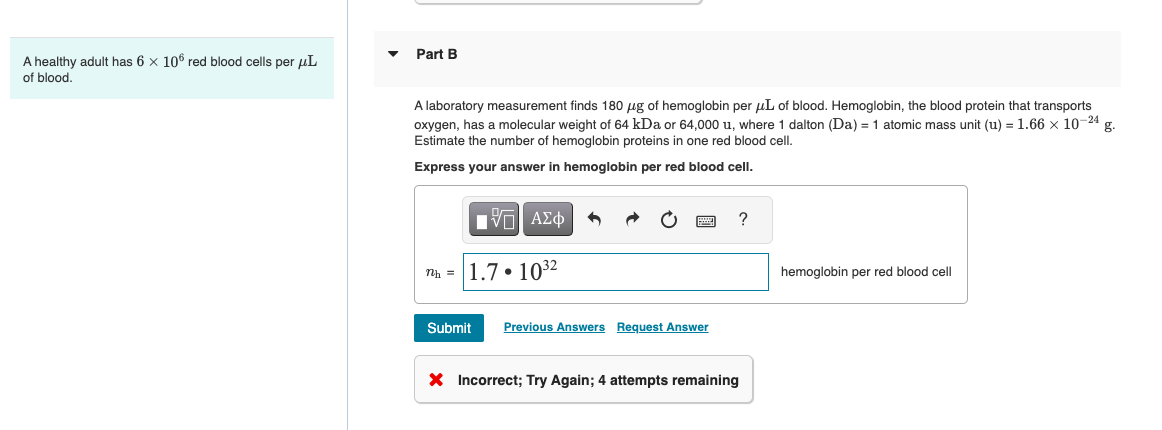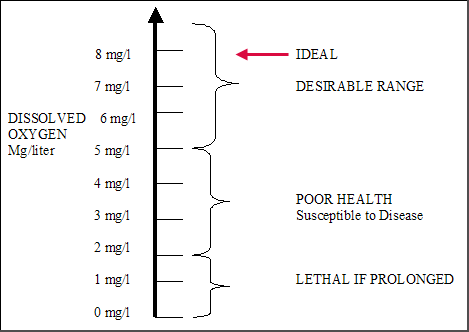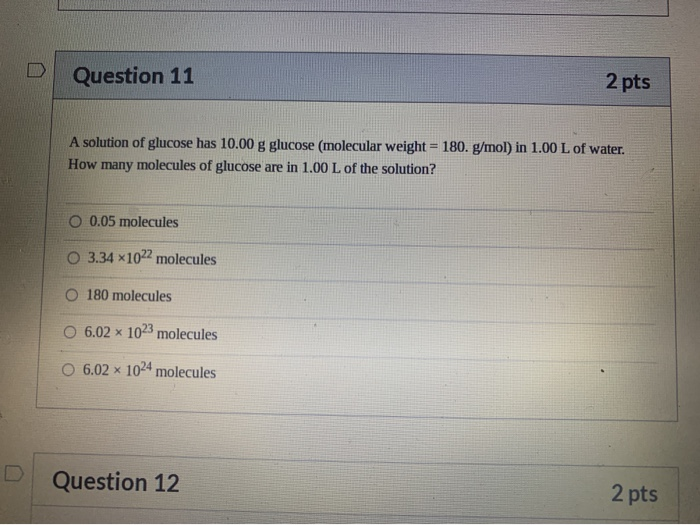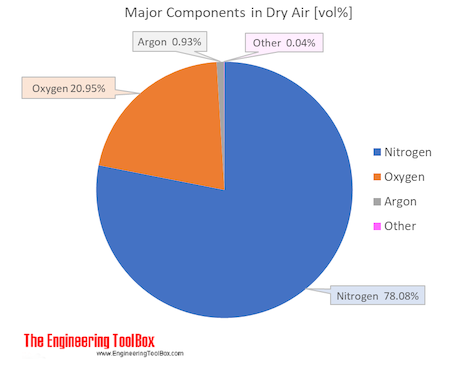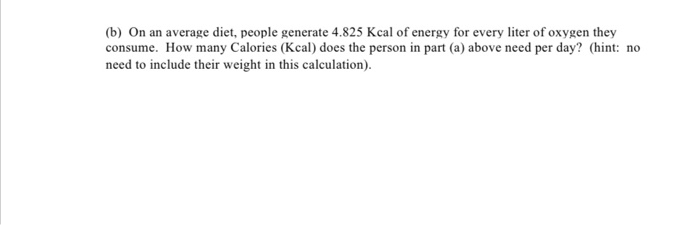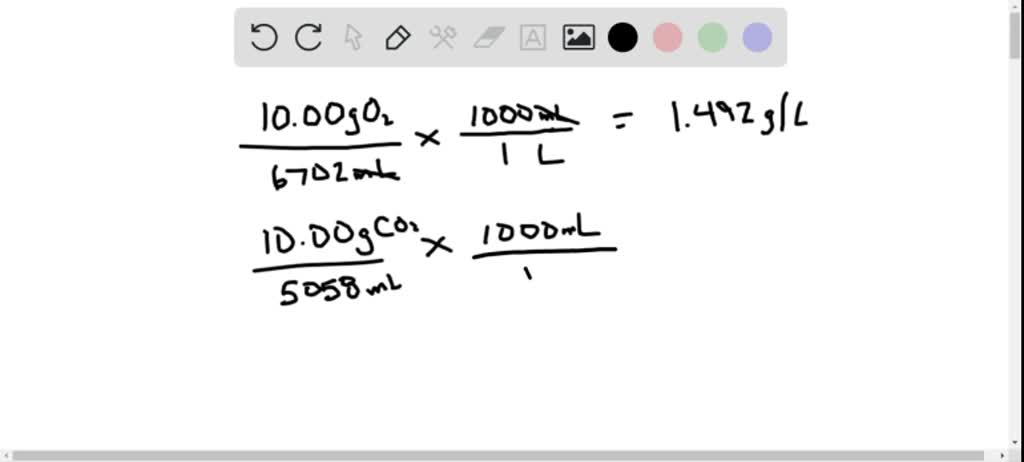
SOLVED:A sample of 10.00 g of oxygen has a volume of 6702 mL The same weight of carbon dioxide occupies 5058 mL (a) What is the density of each gas in g /

SOLVED: SOLUTIONS solution consists of solute dissolved in solvent. A method of expressing solution concentration is in terms of molarity. or moles of solute per liter of solution. mole of compound is

SOLVED: What is the weight of 100.0 sample of SiF4 at STP? A sample of hydrogen gas has volume of 8.56 at a temperature of 0PC and of 1.5 atm: pressure How